Zebra's Industry Solutions can give your business a competitive advantage by connecting people, assets and data to help you make better decisions.
Scale and energise your retail strategy with a digital backbone that unifies your team, informs priorities and drives results with Zebra's retail technology solutions.
Zebra's healthcare technology solutions provide patient identity management, mobile health devices, and business intelligence data to improve efficiency.
Zebra’s manufacturing technology solutions enable manufacturers to become more agile, optimize plant floor performance and embrace market changes.
Zebra's market-leading solutions and products improve customer satisfaction with a lower cost per interaction by keeping service representatives connected with colleagues, customers, management and the tools they use to satisfy customers across the supply chain.
In today's world, the demands on transportation and logistics companies are higher than ever. Dedicated Warehouse, Fleet and Delivery, and Yard and Terminal solutions enable visibility to every aspect of your business and keep operations running flawlessly around the clock.
Zebra's hospitality technology solutions equip your hotel and restaurant staff to deliver superior customer and guest service through inventory tracking and more.
Empower your field workers with purpose-driven mobile technology solutions to help them capture and share critical data in any environment.
Technology is a key enabler in helping the Public Sector provide their frontline and back office workers automate business process and assets with a digital voice.
Zebra’s mobile computing, scanning, and printing solutions connect each operational area in your warehouse to give you the agility to realize transformational gains.
Zebra's range of mobile computers equip your workforce with the devices they need from handhelds and tablets to wearables and vehicle-mounted computers.
Zebra's desktop, mobile, industrial, and portable printers for barcode labels, receipts, RFID tags and cards give you smarter ways to track and manage assets.
Zebra's 1D and 2D corded and cordless barcode scanners anticipate any scanning challenge in a variety of environments, whether retail, healthcare, T&L or manufacturing.
Zebra's extensive range of RAIN RFID readers, antennas, and printers give you consistent and accurate tracking.
Choose Zebra's reliable barcode, RFID and card supplies carefully selected to ensure high performance, print quality, durability and readability.
Zebra's location technologies provide real-time tracking for your organisation to better manage and optimise your critical assets and create more efficient workflows.
Zebra's rugged tablets and 2-in-1 laptops are thin and lightweight, yet rugged to work wherever you do on familiar and easy-to-use Windows or Android OS.
With Zebra's family of fixed industrial scanners and machine vision technologies, you can tailor your solutions to your environment and applications.
Discover Zebra’s range of accessories from chargers, communication cables to cases to help you customise your mobile device for optimal efficiency.
Zebra's OEM scan engines, imagers, and private label OEM products offer flexible integration and help enhance product development with modern OEM technology.
Zebra's environmental sensors monitor temperature-sensitive products, offering data insights on environmental conditions across industry applications.
Keep labor costs low, your talent happy and your organization compliant. Create an agile operation that can navigate unexpected schedule changes and customer demand to drive sales, satisfy customers and improve your bottom line.
Empower the front line with prioritized task notification and enhanced communication capabilities for easier collaboration and more efficient task execution.
Get full visibility of your inventory and automatically pinpoint leaks across all channels.
Reduce uncertainty when you anticipate market volatility. Predict, plan and stay agile to align inventory with shifting demand.
Drive down costs while driving up employee, security, and network performance with software designed to enhance Zebra's wireless infrastructure and mobile solutions.
Explore Zebra’s printer software to integrate, manage and monitor printers easily, maximizing IT resources and minimizing down time.
Make the most of every stage of your scanning journey from deployment to optimization. Zebra's barcode scanner software lets you keep devices current and adapt them to your business needs for a stronger ROI across the full lifecycle.
RFID development, demonstration and production software and utilities help you build and manage your RFID deployments more efficiently.
RFID development, demonstration and production software and utilities help you build and manage your RFID deployments more efficiently.
Zebra DNA is the industry’s broadest suite of enterprise software that delivers an ideal experience for all during the entire lifetime of every Zebra device.
Advance your digital transformation and execute your strategic plans with the help of the right location and tracking technology.
Aurora Focus™ runs on Zebra’s fixed industrial scanners and VS20/VS40/VS70 smart cameras and comes ready-made for specific tasks like barcode reading and verification, OCR, and presence/absence vision inspection.
Zebra Aurora Focus brings a new level of simplicity to controlling enterprise-wide manufacturing and logistics automation solutions. With this powerful interface, it’s easy to set up, deploy and run Zebra’s Fixed Industrial Scanners and Machine Vision Smart Cameras, eliminating the need for different tools and reducing training and deployment time.
Aurora Imaging Library™, formerly Matrox Imaging Library, machine-vision software development kit (SDK) has a deep collection of tools for image capture, processing, analysis, annotation, display, and archiving. Code-level customization starts here.
Aurora Design Assistant™, formerly Matrox Design Assistant, integrated development environment (IDE) is a flowchart-based platform for building machine vision applications, with templates to speed up development and bring solutions online quicker.
Designed for experienced programmers proficient in vision applications, Aurora Vision Library provides the same sophisticated functionality as our Aurora Vision Studio software but presented in programming language.
Aimed at machine and computer vision engineers, Aurora Vision Studio software enables users to quickly create, integrate and monitor powerful machine vision applications without the need to write a single line of code.
Adding innovative tech is critical to your success, but it can be complex and disruptive. Professional Services help you accelerate adoption, and maximise productivity without affecting your workflows, business processes and finances.
Zebra's Managed Service delivers worry-free device management to ensure ultimate uptime for your Zebra Mobile Computers and Printers via dedicated experts.
Find ways you can contact Zebra Technologies’ Support, including Email and Chat, ask a technical question or initiate a Repair Request.
Zebra's Circular Economy Program helps you manage today’s challenges and plan for tomorrow with smart solutions that are good for your budget and the environment.
Success Stories
From improving delivery times and accuracy to offering visibility into device health, or efficiency in your hospital, Zebra provides solutions and products that successfully tackle daily business challenges. But don’t take our word for it. Check out these success stories.

See Zebra Solutions in Action

KW-Kitchenware Achieves 99% Stock Accuracy with Optimized E-commerce Operations
More

Nhat Tin Logistics Elevates Efficiency and Accuracy with Advanced Scanning Solutions
More

Walgreens Boosts Efficiency and Sales with Zebra's Workcloud Actionable Intelligence
More

Iperal Streamlines the Shopping Experience with Scan As You Shop System
More

Avant Improves Customer Satisfaction By Eliminating Picking and Packing Errors
More

Waytek Improves Throughput by 25% with Zebra Autonomous Mobile Robots
More

G3 Boats Achieves "Google Maps" Style Visibility with RFID Technology
More

DPD Germany Improves Efficiency, Productivity and Transparency in Parcel Deliveries
More

Rashideen Egypt for Trade Boosts Sales Performance by 21%
More

Keells Reduces Customer Wait Times with Zebra's Personal Shopper Devices
More

Blain's Farm and Fleet Enhances Efficiency with Zebra Workcloud™ Solutions
More

MPREIS Rises to Meet New Customer Expectations
More

Autodemolizione Pollini Decreases Interruptions by 20%
More

The Works Enhances Stock Management Efficiency with Workcloud Inventory Visibility
More

The Twig Increases Product Visibility and Improves Inventory Management
More

Office Depot Creates Frictionless Omnichannel Shopping Experience
More

Office Depot Leverages Zebra's Solutions to Meet Customer Expectations
More

Office Depot Increases Task Completion Rate by 42% with Zebra Solutions
More

Blue Dart Streamlines Operations and Improves Efficiency with Innovative Solutions
More

Lusqtoff Improves Inventory Visibility and Staff Productivity
More

JAS Worldwide Improves Picking Efficiency with Autonomous Mobile Robots
More

Junior Achievement of South Florida Boosts Interactive Learning for Students
More

Pontifical Catholic University of Ecuador Saves Time, Money and Reduces Error
More

Automated Vision System Helps Hydram Sheet Metalwork Optimize Inspection Process
More

Hippo Harvest Elevates Farming Efficiency with Autonomous Mobile Robots
More

MOSAIC Refines Quality Assessment with Vision System
More

Machine Learning Technology Helps STIHL Cut Cost, Time and Human Error
More

EIT Logistica Sees 40% Improvement in Warehouse Productivity
More
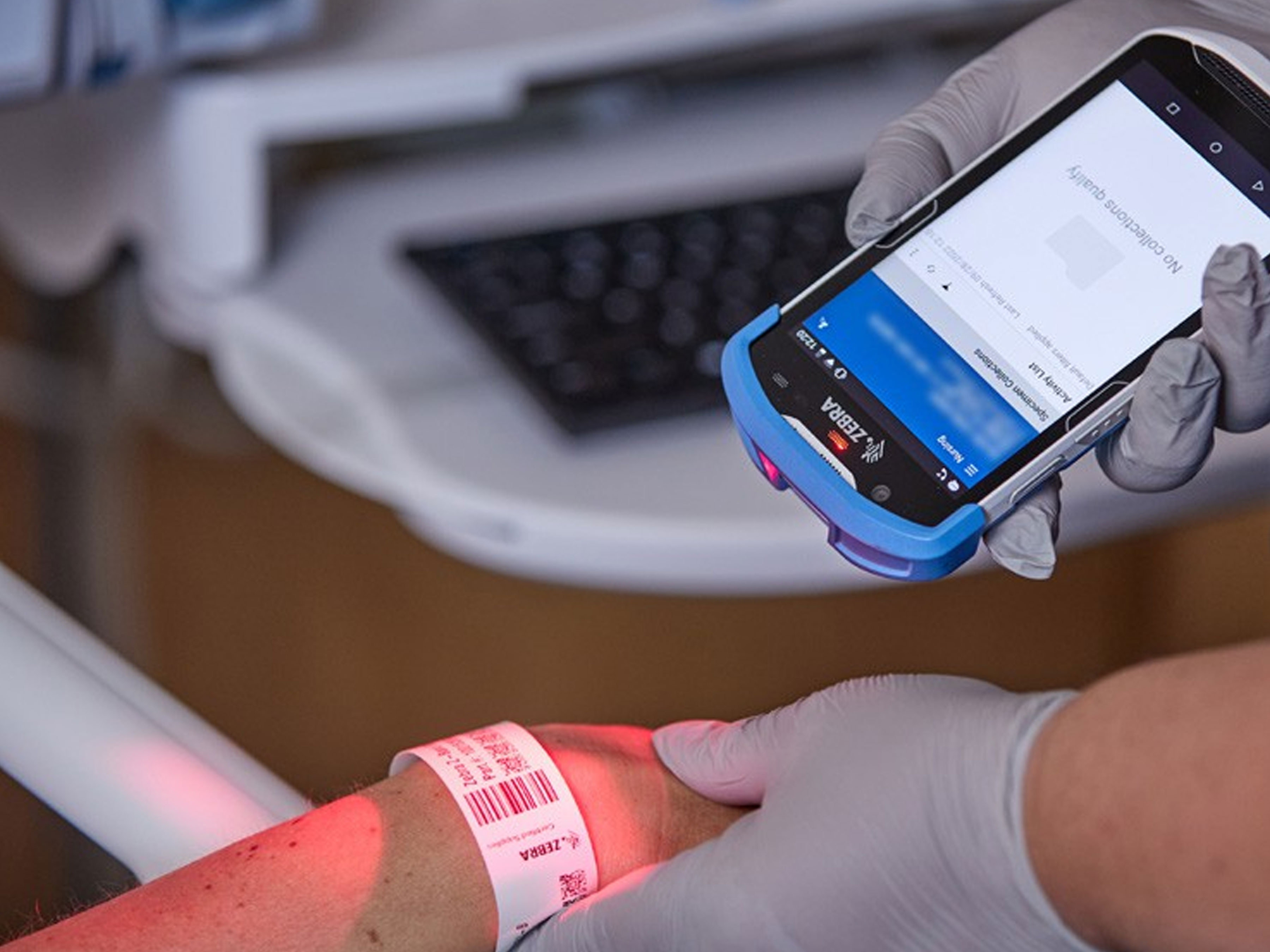
Banner Health Elevates Care and Accelerates Treatment
More

Officeworks Improves Reliability of Payment Processing
More

Le Biscuit Boosts its Omnichannel Operations
More

KINE Robotics Reduces Costs with Accurate Conveyor Tracking System
More

Marexi Marine Technology Sorts Tuna With Accuracy Rates Approaching 100%
More

Elisabeth-TweeSteden Hospital Optimizes Patient Care, Reduces Administrative Tasks
More

Bimbo Bakeries USA Improves Order Accuracy, Minimizes Waste
More

Hull University Teaching Hospital Improves Workflows with Real-Time Visibility of Assets
More

Zebra Technologies Keeps Pace with Demands at its Heerenveen Warehouse
More

Dorman Products Reduces Travel Waste with Autonomous Mobile Robots
More

Austin Lighthouse Empowers Employees, Doubles Productivity
More

Zebra's Reflexis Task Management Solutions Help Farmacity Meet Customer Expectations
More

CVS Health Drives Efficiencies with Zebra's Reflexis Software Solutions
More

Rockler Woodworking Streamlines Communication with Reflexis
More

Ecoembes Helps Reduce Plastic Waste Through Smart Technology
More
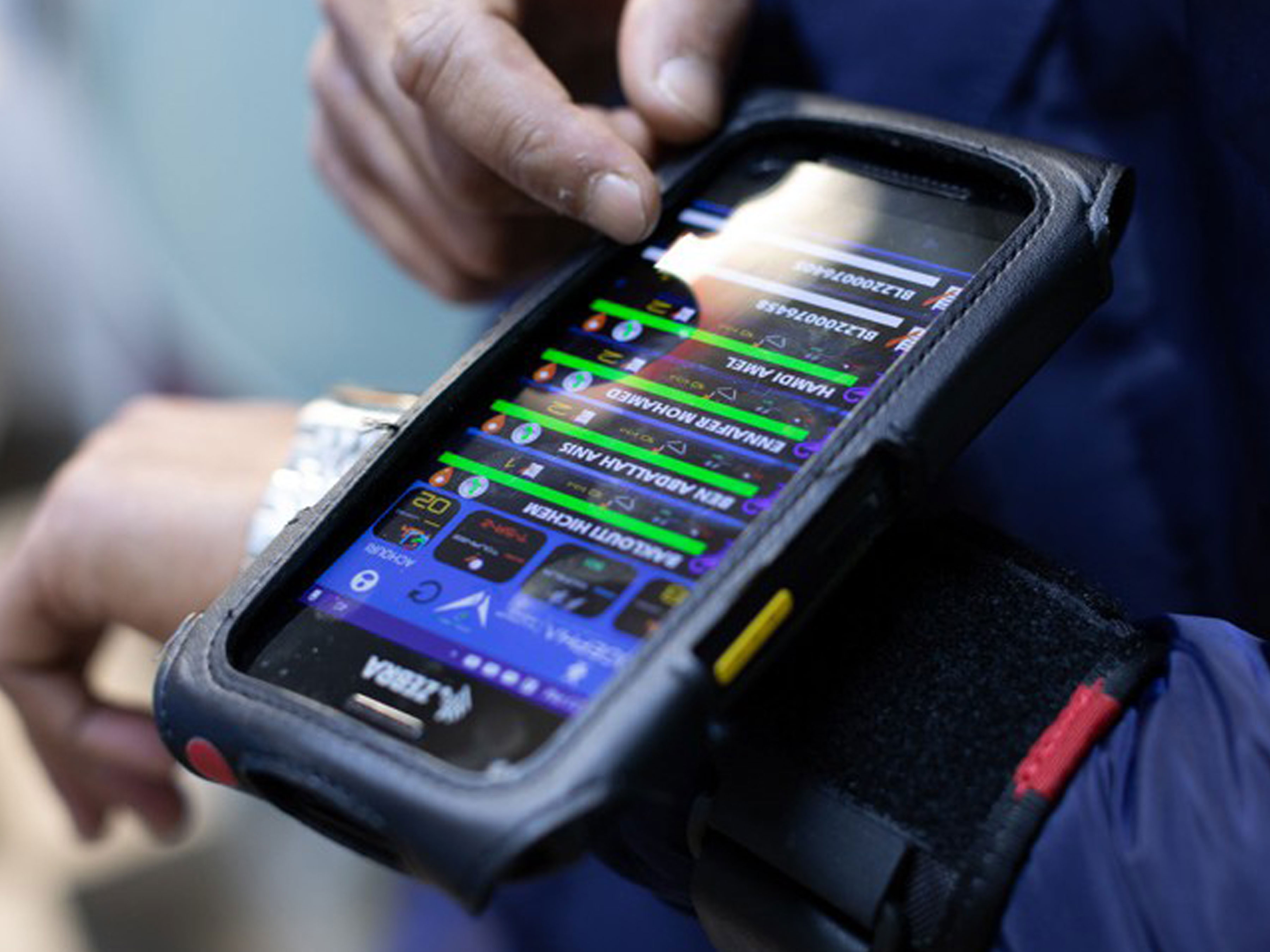
Cogepha Improves Speed and Transparency of Operations
More

Qore Creates An Automated Solution For Issuing Bank Cards
More

Alpro Pharmacy Boosts Productivity and Accuracy Amid Substantial Growth
More

DynaEnergetics Boosts Inventory Visibility with Zebra Mobile Android™ Solution
More

Liverpool Gains Real-Time Visibility Into Projects and Tasks
More

Hunt Country Components Improves Customer Satisfaction With RFID Tracking
More

Kesko Reduces Costs, Boosts Efficiency With Enterprise Grade Print Solution
More

Mobile Solution Helps Cargus Improve Transparency and Enhance Customer Service
More

Foodstuffs South Island Improves Efficiency With Zebra Handheld Devices
More

Casar Streamlines Tomato Distribution With Zebra RFID
More

Bosch Further Automates Inspection Process With Enhanced Vision System
More

Cyprus Accelerates Visitor Checks At Transit Hubs, Public Places
More

Defense Organization Finds Reliable Solution To Withstand Tough Environments
More

QE Facilities Enhances Reliability With Specimen Tracking Solution
More

Fanatics Streamlines Communication, Improves Task Visibility
More

Paracelsus-Kliniken Digitalizes Complex Hospital Processes
More

Office Depot Enhances Its Supply Chain Operations
More

Lowes Foods Reduces Total Retail Loss with Zebra Prescriptive Analytics
More

Murchison Defiba Gains Visibility, Peace of Mind With Mobile Solution
More

Waterman Onions Adopts Digital Solution To Streamline Operations
More

Shoe Sensation Optimizes Communication and Scheduling With Reflexis
More

The Noerpel Group Accelerates Goods Receipt and Onward Handling
More

MNG Kargo Boosts Handling Capacity to Meet Increased Demand
More

Transforming the Grocery Experience for Today’s Digital-Savvy Shoppers
More

Construction Material Distributor Digitalizes Workflow
More

Philadelphia Automates Tracking Of Voting Equipment And Materials
More

Packaging Manufacturer Improves Traceability With Seamless Transition to Android™
More
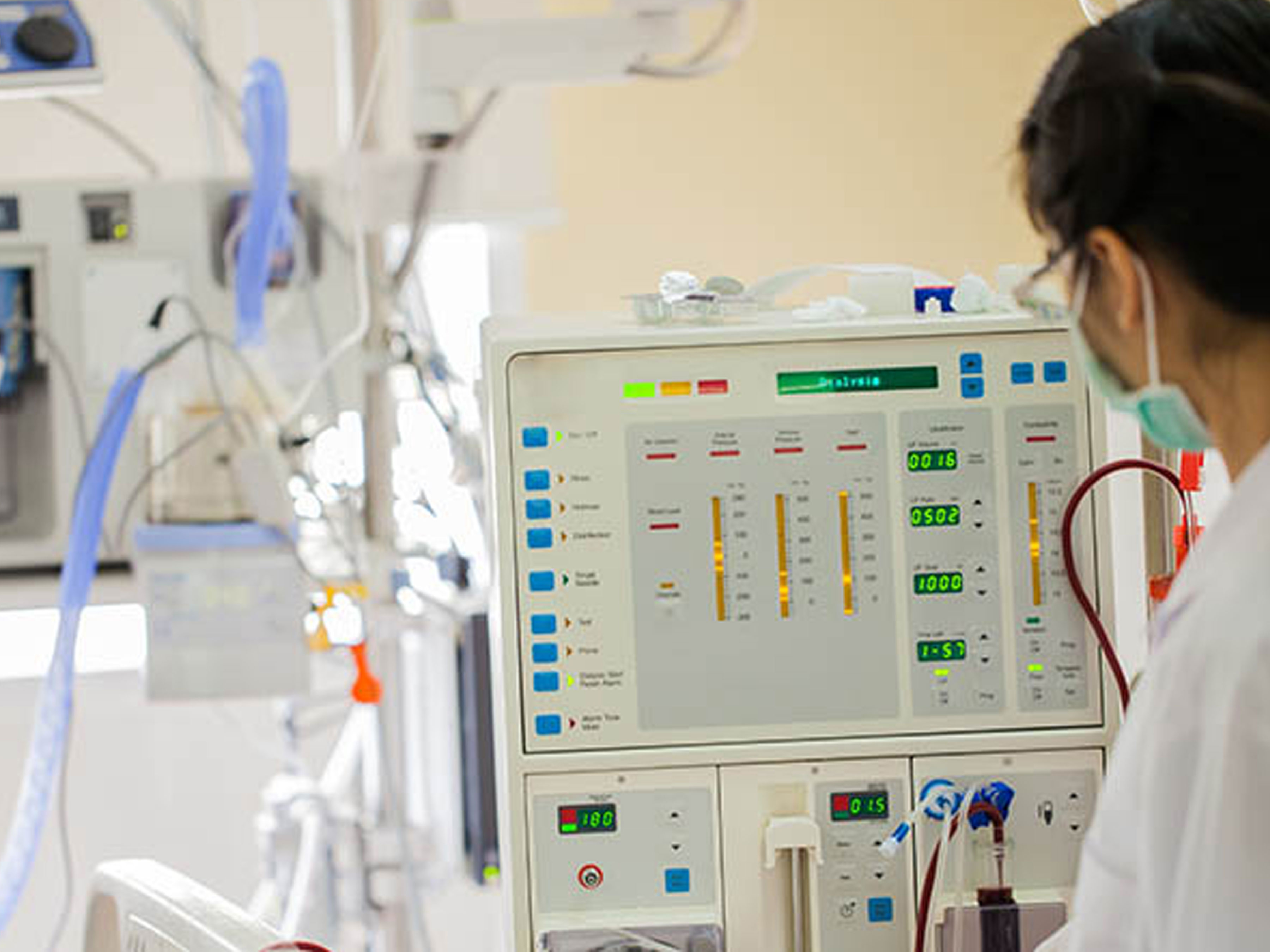
Redbite
More

Be Switchcraft Sees Manufacturing Process In Real Time With Rfid
More

Veritas Increases Efficiency of Meter Replacement Process
More

Raja Group Digitalizes Its Warehouse Process with the Help of Zebra's High-performance Technology Solutions
More

TrainOSE Digitalizes Ticketing System
More

Vera Bradley
More

Multicenter
More

Vera Bradley
More

Altru Specialty Pharmacy
More

OmegaFlex
More

Lowe's Turns to New Tech to Manage Flow of Goods
More

Multicenter
More

CEWE Speeds Up Production with Industrial Scanners
More

Deutsche Post DHL Makes Big Move to Improve Deliveries
More

Hasenauer+Koch GmbH + Co. KG
More

Multimoto
More

Stuart Logistics Improves Sorting Center Operations
More

Lowe’s Digital Transformation Streamlines Operations and Bolsters Omnichannel Capabilities
More

RevLogical
More

Forza Cash Logistics Uses Zebra Handheld Mobile Computers to Provide Real-time Updates and Reduce Errors While Transporting Valuables
More

Calderdale and Huddersfield NHS Foundation Trust
More
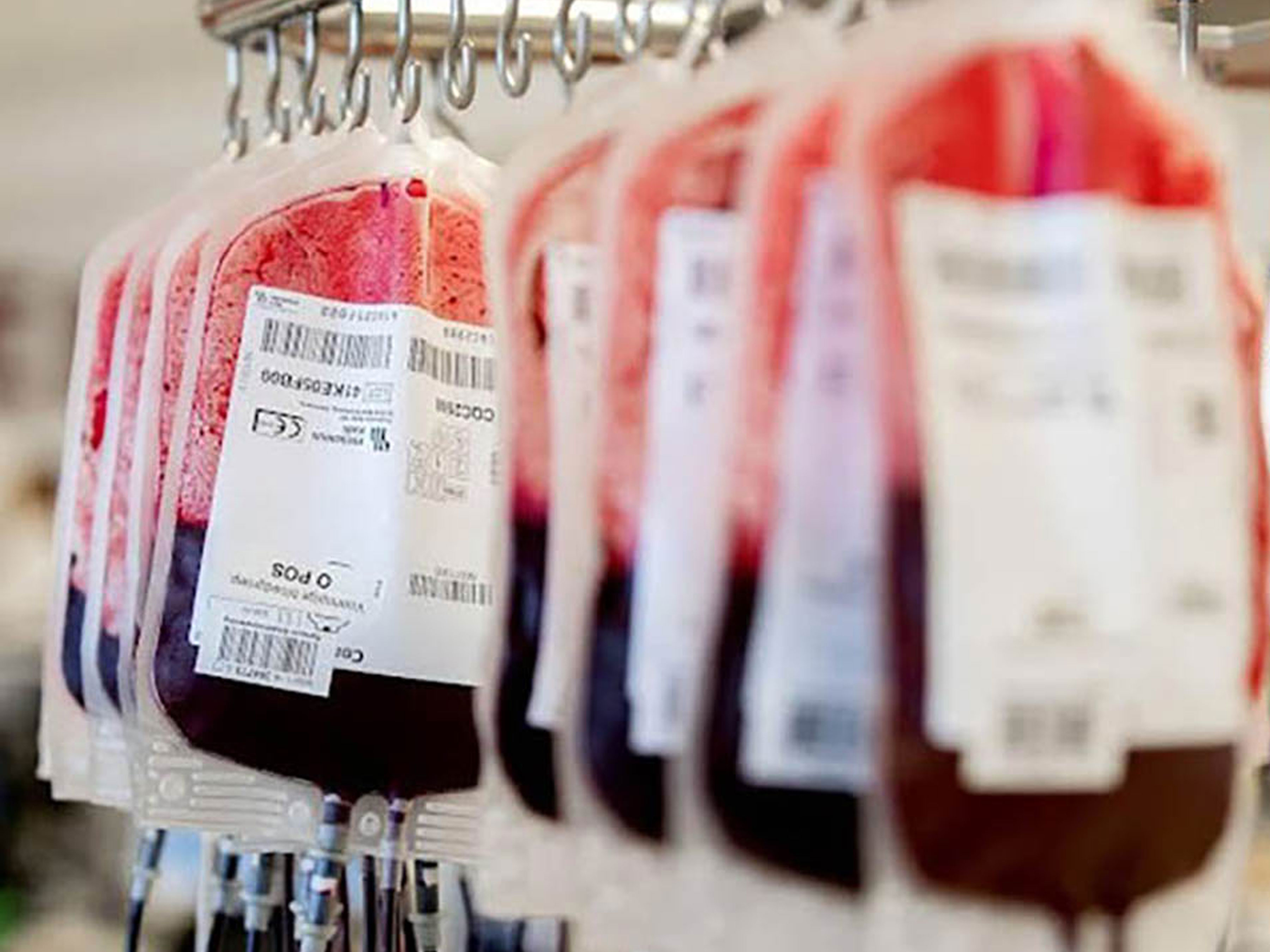
Sanquin
More

Teatr Wielki
More

Computacenter
More

Toyota Material Handling Uses Zebra MotionWorks Enterprise to Track and Streamline Product Movements in Real Time
More

Nicholasville Fire Department
More

Waitrose
More

Waitrose
More

Vera Bradley
More
Siemens Healthineers
More

Carol Pharmacies
More
Crystal
More

Havan
More

Fashionalia
More

Truman Medical Centers/ Cerner Corporation
More

St George, Utah
More

The Royal Children’s Hospital Improves Patient Safety With Accurate Scanning Of Blood Products
More

K. Hansen Transport
More

Comercial Kywi
More

Dinet Boosts Warehouse Efficiencies by 20%
More

Dutch Valley Food Distributors Solves Picking Challenges
More

IFD Capita
More

Swiss Medical Group
More

McDonald's Brazil Uses Tablets to Speed Up Drive-Thru Queues
More

Renault
More

Wendy's
More

Walgreens
More

Orica
More

Autotodo Mexicana
More

Hans Anders
More

Northern Lakes Fire District
More

Sogegross Group
More

Northshore Care Supply
More

BetXS
More

ArcBest
More

De Vries Transport Hallum
More

Oegema Transport
More

Caesars Entertainment
More

Leroy Merlin
More

Mt Buller Alpine
More

Iperal
More

Louis Widmer
More

Lithuania Post
More

jem & fix
More
Pasco County Fire Rescue
More

EDA Neptune
More

AFV Acciaierie Beltrame
More

Saddle Creek Logistics Services
More

Whirlpool Corporation
More

Ford Motor Company
More

Bradford Airport Logistics
More

FITT Increases Value of WMS with Mobile Solution
More

Kaufland
More

DRMP Enhances Asset Management with RFID Technology
More

Wavin
More

Bonita Springs Fire Control and Rescue District
More

Kimbark Laundry & Dry Cleaning
More

Direct Relief
More

Sally Beauty Holdings Streamlines Store Operations With Reflexis Solutions
More
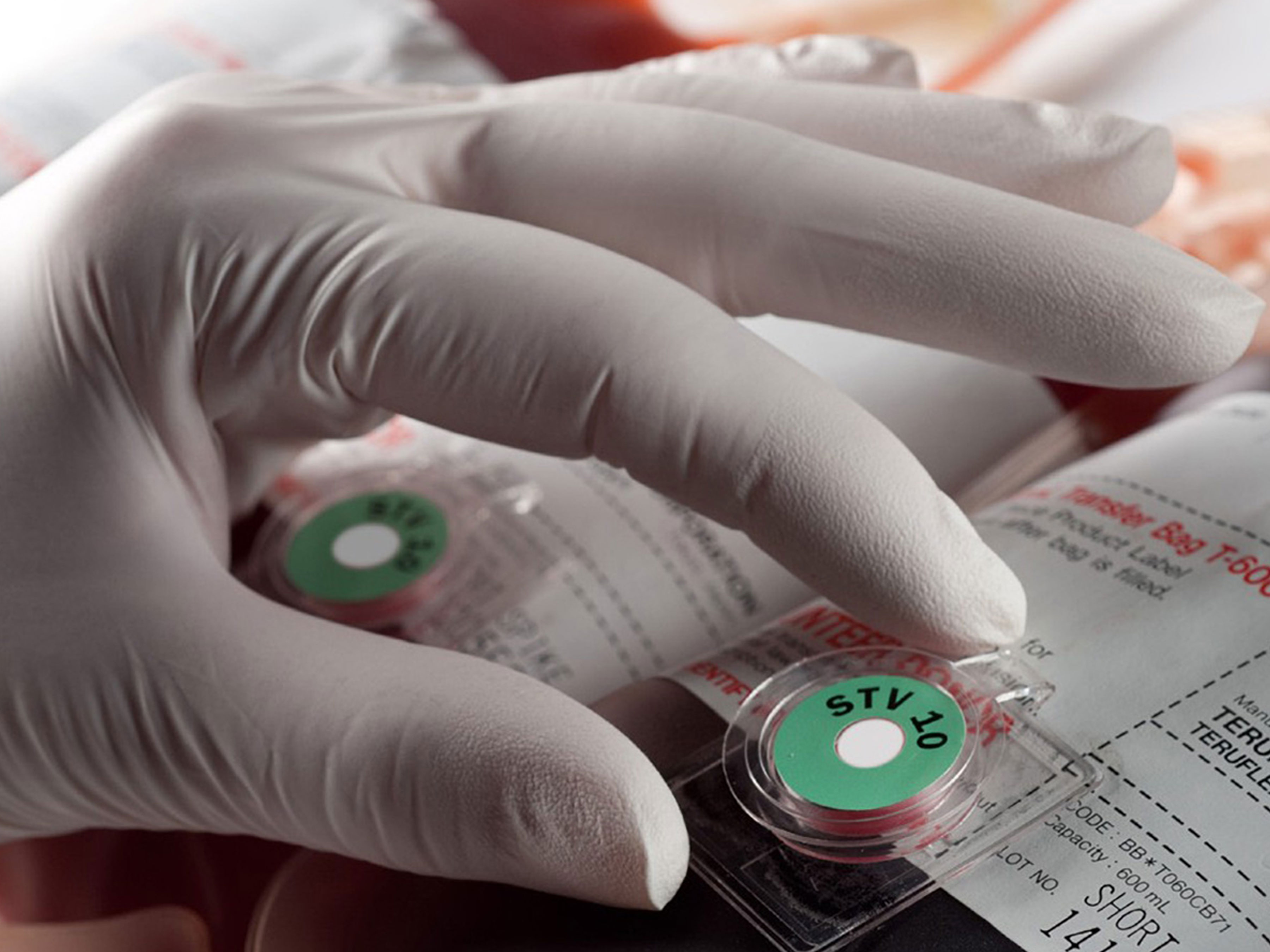
Temperature Indicators Help University Health Kansas City Monitor Blood Bags
More

Cardiovascular Foundation of Colombia Optimizes Traceability, Improves Patient Care
More

TransTracker Indicators Help AON Pharmacy Deliver With Confidence
More

ID Logistics Polska Boosts Operations With Latest Warehousing Technology
More
Learn More About Zebra's Customer Advocacy Program
Legal Terms of Use Privacy Policy Supply Chain Transparency
ZEBRA and the stylized Zebra head are trademarks of Zebra Technologies Corp., registered in many jurisdictions worldwide. All other trademarks are the property of their respective owners. ©2024 Zebra Technologies Corp. and/or its affiliates.

