Transform retail operations with Zebra’s retail technology solutions, featuring hardware and software for improving inventory management and empowering teams.
Streamline operations with Zebra’s healthcare technology solutions, featuring hardware and software to improve staff collaboration and optimize workflows.
Enhance processes with Zebra’s manufacturing technology solutions, featuring hardware and software for automation, data analysis, and factory connectivity.
Zebra’s transportation and logistics technology solutions feature hardware and software for enhancing route planning, visibility, and automating processes.
Learn how Zebra's public sector technology solutions empower state and local governments to improve efficiency with asset tracking and data capture devices.
Zebra's hospitality technology solutions equip your hotel and restaurant staff to deliver superior customer and guest service through inventory tracking and more.
Zebra's market-leading solutions and products improve customer satisfaction with a lower cost per interaction by keeping service representatives connected with colleagues, customers, management and the tools they use to satisfy customers across the supply chain.
Empower your field workers with purpose-driven mobile technology solutions to help them capture and share critical data in any environment.
Zebra's range of Banking technology solutions enables banks to minimize costs and to increase revenue throughout their branch network. Learn more.
Zebra's range of mobile computers equip your workforce with the devices they need from handhelds and tablets to wearables and vehicle-mounted computers.
Zebra's desktop, mobile, industrial, and portable printers for barcode labels, receipts, RFID tags and cards give you smarter ways to track and manage assets.
Zebra's 1D and 2D corded and cordless barcode scanners anticipate any scanning challenge in a variety of environments, whether retail, healthcare, T&L or manufacturing.
Zebra's extensive range of RAIN RFID readers, antennas, and printers give you consistent and accurate tracking.
Choose Zebra's reliable barcode, RFID and card supplies carefully selected to ensure high performance, print quality, durability and readability.
Zebra's location technologies provide real-time tracking for your organization to better manage and optimize your critical assets and create more efficient workflows.
Zebra's rugged tablets and 2-in-1 laptops are thin and lightweight, yet rugged to work wherever you do on familiar and easy-to-use Windows or Android OS.
With Zebra's family of fixed industrial scanners and machine vision technologies, you can tailor your solutions to your environment and applications.
Zebra’s line of kiosks can meet any self-service or digital signage need, from checking prices and stock on an in-aisle store kiosk to fully-featured kiosks that can be deployed on the wall, counter, desktop or floor in a retail store, hotel, airport check-in gate, physician’s office, local government office and more.
Adapt to market shifts, enhance worker productivity and secure long-term growth with AMRs. Deploy, redeploy and optimize autonomous mobile robots with ease.
Discover Zebra’s range of accessories from chargers, communication cables to cases to help you customize your mobile device for optimal efficiency.
Zebra's environmental sensors monitor temperature-sensitive products, offering data insights on environmental conditions across industry applications.
Enhance frontline operations with Zebra’s AI software solutions, which optimize workflows, streamline processes, and simplify tasks for improved business outcomes.
Zebra Workcloud, enterprise software solutions boost efficiency, cut costs, improve inventory management, simplify communication and optimize resources.
Keep labor costs low, your talent happy and your organization compliant. Create an agile operation that can navigate unexpected schedule changes and customer demand to drive sales, satisfy customers and improve your bottom line.
Drive successful enterprise collaboration with prioritized task notifications and improved communication capabilities for easier team collaboration.
Get full visibility of your inventory and automatically pinpoint leaks across all channels.
Reduce uncertainty when you anticipate market volatility. Predict, plan and stay agile to align inventory with shifting demand.
Drive down costs while driving up employee, security, and network performance with software designed to enhance Zebra's wireless infrastructure and mobile solutions.
Explore Zebra’s printer software to integrate, manage and monitor printers easily, maximizing IT resources and minimizing down time.
Make the most of every stage of your scanning journey from deployment to optimization. Zebra's barcode scanner software lets you keep devices current and adapt them to your business needs for a stronger ROI across the full lifecycle.
RFID development, demonstration and production software and utilities help you build and manage your RFID deployments more efficiently.
RFID development, demonstration and production software and utilities help you build and manage your RFID deployments more efficiently.
Zebra DNA is the industry’s broadest suite of enterprise software that delivers an ideal experience for all during the entire lifetime of every Zebra device.
Advance your digital transformation and execute your strategic plans with the help of the right location and tracking technology.
Boost warehouse and manufacturing operations with Symmetry, an AMR software for fleet management of Autonomous Mobile Robots and streamlined automation workflows.
The Zebra Aurora suite of machine vision software enables users to solve their track-and-trace, vision inspection and industrial automation needs.
Zebra Aurora Focus brings a new level of simplicity to controlling enterprise-wide manufacturing and logistics automation solutions. With this powerful interface, it’s easy to set up, deploy and run Zebra’s Fixed Industrial Scanners and Machine Vision Smart Cameras, eliminating the need for different tools and reducing training and deployment time.
Aurora Imaging Library™, formerly Matrox Imaging Library, machine-vision software development kit (SDK) has a deep collection of tools for image capture, processing, analysis, annotation, display, and archiving. Code-level customization starts here.
Aurora Design Assistant™, formerly Matrox Design Assistant, integrated development environment (IDE) is a flowchart-based platform for building machine vision applications, with templates to speed up development and bring solutions online quicker.
Designed for experienced programmers proficient in vision applications, Aurora Vision Library provides the same sophisticated functionality as our Aurora Vision Studio software but presented in programming language.
Aurora Vision Studio, an image processing software for machine & computer vision engineers, allows quick creation, integration & monitoring of powerful OEM vision applications.
Adding innovative tech is critical to your success, but it can be complex and disruptive. Professional Services help you accelerate adoption, and maximize productivity without affecting your workflows, business processes and finances.
Zebra's Managed Service delivers worry-free device management to ensure ultimate uptime for your Zebra Mobile Computers and Printers via dedicated experts.
Find ways you can contact Zebra Technologies’ Support, including Email and Chat, ask a technical question or initiate a Repair Request.
Zebra's Circular Economy Program helps you manage today’s challenges and plan for tomorrow with smart solutions that are good for your budget and the environment.
The Zebra Knowledge Center provides learning expertise that can be tailored to meet the specific needs of your environment.
Zebra has a wide variety of courses to train you and your staff, ranging from scheduled sessions to remote offerings as well as custom tailored to your specific needs.
Build your reputation with Zebra's certification offerings. Zebra offers a variety of options that can help you progress your career path forward.
Build your reputation with Zebra's certification offerings. Zebra offers a variety of options that can help you progress your career path forward.
Stories from the Edge: Maintaining Medication Temperature is Key to Patient Treatment
Did you know that, like the human body, certain medications can become less effective if they are exposed to extreme temperatures? It’s true, particularly when they get too hot or too cold. Medications that have been exposed to temperatures outside the range recommended by their manufacturers may not work as well as they should. That is why Preveon Specialty Pharmacy emphasizes controlling and monitoring the temperature of the highly sensitive biologics that it handles and asks patients to do the same.
Fevers Aren’t the Only “Temperature” Concern and Over-the-Counter Thermometers Aren’t the Only Way to Monitor Temperatures
Preveon may fill prescription medications, but it is not your typical pharmacy. It is a specialty pharmacy that focuses on high-cost, high-touch medication therapy for patients with complex disease states. Medications in the specialty pharmacy’s formulary range from oral to cutting edge injectable and biologic products. As such, Preveon takes an active and holistic role in managing patients’ treatment protocols in partnership with primary care teams.
In fact, as part of its unique Disease Therapy Management Program, Preveon clinical pharmacists serve as 24/7 advisors to patients receiving drugs or biologics, closely monitoring their compliance with treatment protocols as well as their bodies’ responses to each drug. They are highly invested in each patient’s success and want to ensure that nothing compromises the potential of a positive outcome, especially not a quality-compromised medication.
Preveon knows that the effectiveness of temperature-sensitive drugs and biologics is dependent on storage at the proper temperature throughout the cold chain, from storage at the pharmacy to handling during shipping. It is also dependent on how patients store their medications after receipt at home. For example, one study found that the majority of patients using biologic disease-modifying antirheumatic drugs (DMARDs) do not store their drugs within the recommended temperature range.
Measuring the temperature of the medication at any one point can confirm the current temperature, but how would you know if the medication was exposed to potentially damaging temperatures during picking, packing, shipping and unpacking? You wouldn’t – without advanced temperature monitoring tools. Thus, the reason why Preveon contacted Temptime Corporation, now a part of Zebra Technologies.
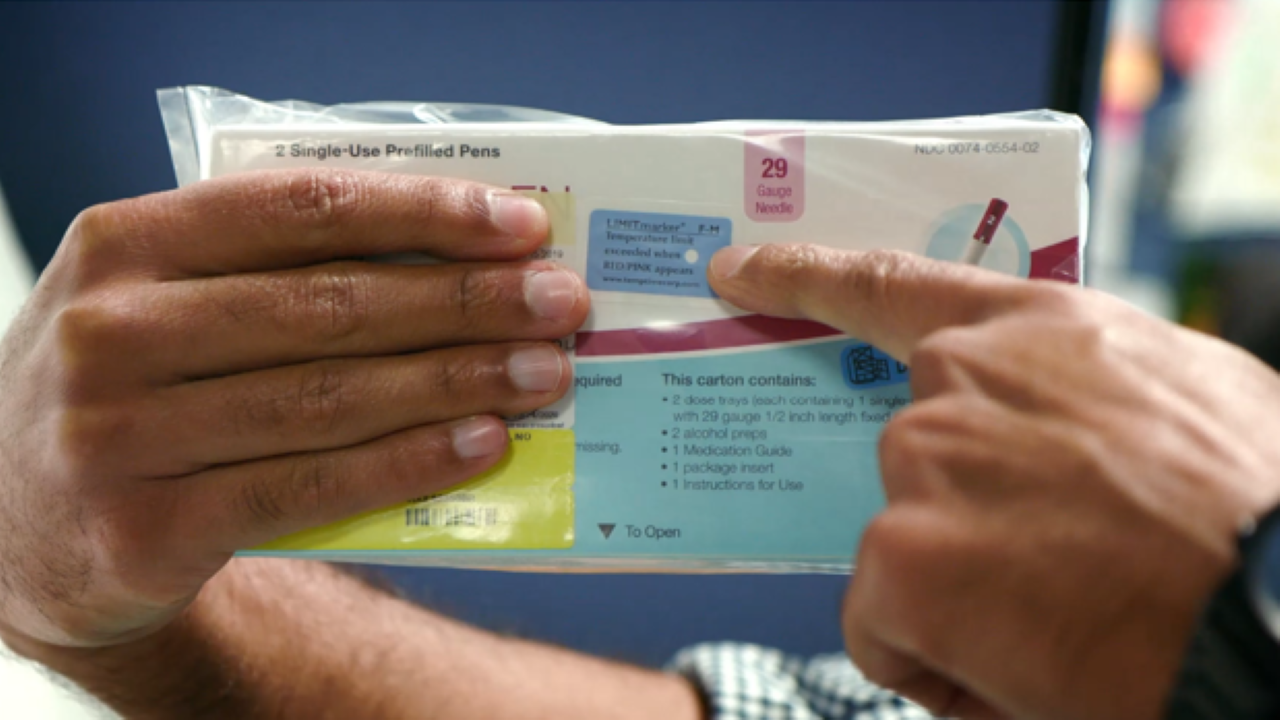
As clinical pharmacist, Phillip Hivale explains in the below video, Preveon wanted to give its pharmacists, order fillers and even patients a super-easy way to confirm whether or not a biologic has been exposed to a potentially damaging temperature level at any point in time. How? By looking at the label.
Check this out:
Integrity is the Priority in Today’s Pharmaceutical Supply Chains
Food safety is always on the minds of producers, warehouse operators, distributors and restaurateurs. Even home cooks and consumers are closely scrutinizing food quality these days. We want to know that the quality of what we’re putting into our bodies, or giving others to put into their bodies, hasn’t been compromised during storage, transport or preparation.
That is why equal scrutiny must be given to pharmaceuticals – partially because of tight government and industry regulations, but mostly because of the correlation between a drug’s potency at the time of administration and its effectiveness. These prescriptions change hands many different times as they travel from the production facility to the “last mile” – whether that is in a clinic or a patient’s home.
Fortunately, a growing number of State Board of Pharmacy regulations and accreditation body standards are encouraging manufacturers, distributors, healthcare providers and specialty pharmacies, such as Preveon, to deploy technologies to demonstrate that the cold chain has been respected during handling. By putting multiple quality control measures in place throughout the cold chain, they can help maintain the potency of medications, particularly temperature-sensitive biologics.
For example, you may find wireless temperature sensors installed in cold storage facilities within manufacturing plants, warehouses, distribution centers and even hospitals and pharmacies to monitor climate conditions. They can even be attached inside the handheld coolers used for pharmacies’ courier delivery services, as you saw in the Preveon video above. However, those devices can’t be practically attached to individual drug packaging and are too expensive to be used in individual shipments to a patient’s home.
That is why color-changing temperature-sensing labels such as the LIMITmarker™ F-M used by Preveon have become so valuable to healthcare providers. They can simply, yet scientifically, indicate that drugs have been exposed to potentially damaging temperatures both prior to and after arrival at hospitals or patients’ homes and subsequently elicit corrective action. Both patients and caregivers have a way of knowing when the temperature control within a shipment has been compromised and may need to be replaced. As a result, they gain confidence in the integrity of these specialty treatments while simultaneously reducing the cost burden associated with “not knowing” about a drug’s quality.
(The average monthly cost of a specialty drug can exceed $5,000. And without the use of a temperature indicator in each shipment, there is an increased risk of the high-value delivery being rejected due to a suspected, but not verified, temperature excursion. The rejected shipment sets into motion a series of actions that result in a product reship and replacement, which is often a cost borne by the specialty pharmacy, and also a delay in the timely administration of the drug. When medications are not administered on time and according to the patient’s unique schedule, patients may experience an immediate increase in symptoms. Eliminating the direct and indirect costs of replacing a compromised shipment has a big impact on the specialty pharmacies’ profitability and the quality of life of the patient!)
In other words, temperature-monitoring technologies are well worth the investment given that peace of mind is priceless – to pharmaceutical manufacturers, distributors, physicians, pharmacists and patients.
###
Editor’s Note:
You can learn about the many different temperature monitoring and sensing technology solutions being used to monitor many different temperature sensitive products, including food, vaccines, pharmaceuticals, and blood on Zebra’s website.

Tony Cecchin
Tony Cecchin leads Zebra’s Global Supplies organization which includes barcode labels, wristbands, RFID medio, etc, as Vice President and General Manager. Tony also serves as President of Temptime which was acquired by Zebra in February 2019. Temptime is a leading manufacturer of temperature sensing labels and monitoring solutions.
Tony is responsible for all aspects of the businesses including strategy, portfolio management, manufacturing and research and development. His passion and energy have enabled a transformation of the traditional Supplies business to include a new category of intelligent supplies. Tony began his career over 30 years ago with Motorola where he led various organizations globally. His deep experience in product management, international business, joint ventures and strategy defined a proven track record of success. Tony recently served as Vice President, Portfolio Management for Zebra’s services organization managing the support and managed services portfolio.
Tony holds a BA and MBA from Loyola University of Chicago and has been awarded three United States patents.
Zebra Developer Blog
Zebra Developer Blog
Are you a Zebra Developer? Find more technical discussions on our Developer Portal blog.
Zebra Story Hub
Zebra Story Hub
Looking for more expert insights? Visit the Zebra Story Hub for more interviews, news, and industry trend analysis.
Search the Blog
Search the Blog
Use the below link to search all of our blog posts.
Most Recent
Legal Terms of Use Privacy Policy Supply Chain Transparency
ZEBRA and the stylized Zebra head are trademarks of Zebra Technologies Corp., registered in many jurisdictions worldwide. All other trademarks are the property of their respective owners. ©2025 Zebra Technologies Corp. and/or its affiliates.






